Introduction
The tumor microenvironment (TME) is the cellular environment in which tumors exist. Some characteristics of the TME include sustained angiogenesis, acidification and a hypoxic condition. In addition, the non-neoplastic cells, which are collectively called stroma, also assist in tumor survival and progression. It has been shown that disrupting the tumor-stromal balance has significant effects on tumor survival, and effective treatment can be achieved by targeting one or more of the stromal components to increase drug penetration. For this purpose, polymer-based delivery system can be developed to take advantage of the characteristics of the TME and allow the selective uptake of anti-tumor drugs.
Synthesis of the Cellax System
A nanoparticle formulation of docetaxel (DTX) was designed in our lab to address the strengths and limitations of current taxane delivery systems: PEGylation, high drug conjugation efficiency (>30 wt %), a slow-release mechanism, and a well-defined and stable nanoparticle identity were identified as critical design parameters. The polymer conjugate was synthesized with carboxymethylcellulose (CMC), an established pharmaceutical excipient characterized by a high density of carboxylate groups permitting increased conjugation of a drug. CMC was chemically modified through acetylation to eliminate its gelling properties and to improve solvent solubility, enabling high yield and reproducible conjugation of DTX and poly(ethylene glycol). (Ernsting et al., 2011; http://pubs.acs.org/doi/abs/10.1021/bc200284b)
Some existing limitations with the conjugation with polysaccharides include low drug loading (<20%), biologically active polymer chemistry (i.e., heparin and hyaluronan), or rapid release (>20–30% per day). Carboxymethylcellulose (CMC) is an attractive candidate polymer to address the existing limitations as a conjugate backbone, as CMC is approved for use in parenteral formulations, is not biologically active like heparin or hyaluronic acid, and has a high degree of substitution of carboxylic acids for drug conjugation, enabling the incorporation of a high drug content. (Ernsting et al., 2012; http://www.sciencedirect.com/science/article/pii/S0142961211012774)
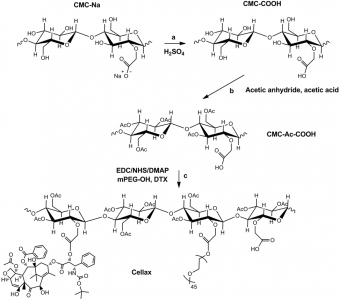
Figure 1. Chemical synthesis of the Cellax polymer conjugate and self-assembly of nanoparticles. (Ernsting et al., 2011; http://pubs.acs.org/doi/abs/10.1021/bc200284b)
Applications
- tumor treatment
k
k
k
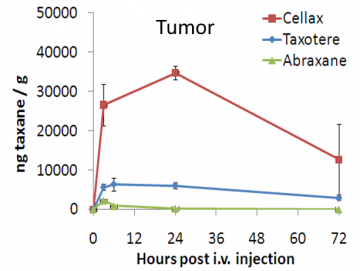
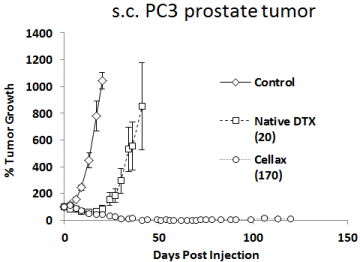
Figure 2. Cellax displays prolonged PK (left) and enhanced anti-tumor efficacy (right) (Hoang et al., 2014 https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4122598/)
- modification of tumor microenvironment
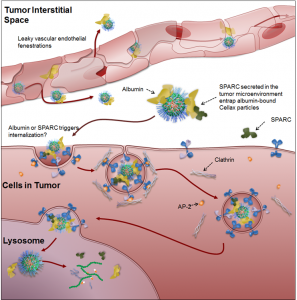
Figure 3. Proposed mechanism of delivery for Cellax nanoparticles. Cellax adsorbs albumin in circulation and is deposited within the tumor interstitium via exploitation of the enhanced permeability and retention (EPR) effect. SPARC produced in the tumor microenvironment binds to the surface albumin on the Cellax nanoparticles and thus traps the particles in the tumor. Cellax is internalized via a clathrin-mediated mechanism and finally ends up in the endolysosomal compartment, where the polymer is broken down and the drug is released.
(Hoang et al. Biomaterials 2015. http://www.sciencedirect.com/science/article/pii/S0142961215003877)
- reduction of metastases
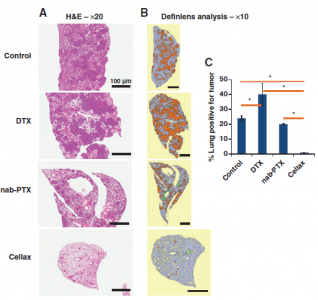
Figure 4. Antimetastatic effect of native docetaxel (DTX), nab-paclitaxel, and Cellax against the 4T1 breast tumor. Lung tissues from the Balb/c mice bearing 4T1 orthotopic breast tumors were analyzed for metastases. H&E-stained sections (A) showed that the prevalence of tumors was visibly reduced in Cellax-treated mice. The tissue sections were analyzed in Definiens software (B) and the quantitative analysis (C) showed that tumor burden (orange) as a fraction of lung tissue (blue) was significantly reduced in the Cellax-treated mice.
(Murakami, Ernsting et al., 2013. http://cancerres.aacrjournals.org/content/73/15/4862)